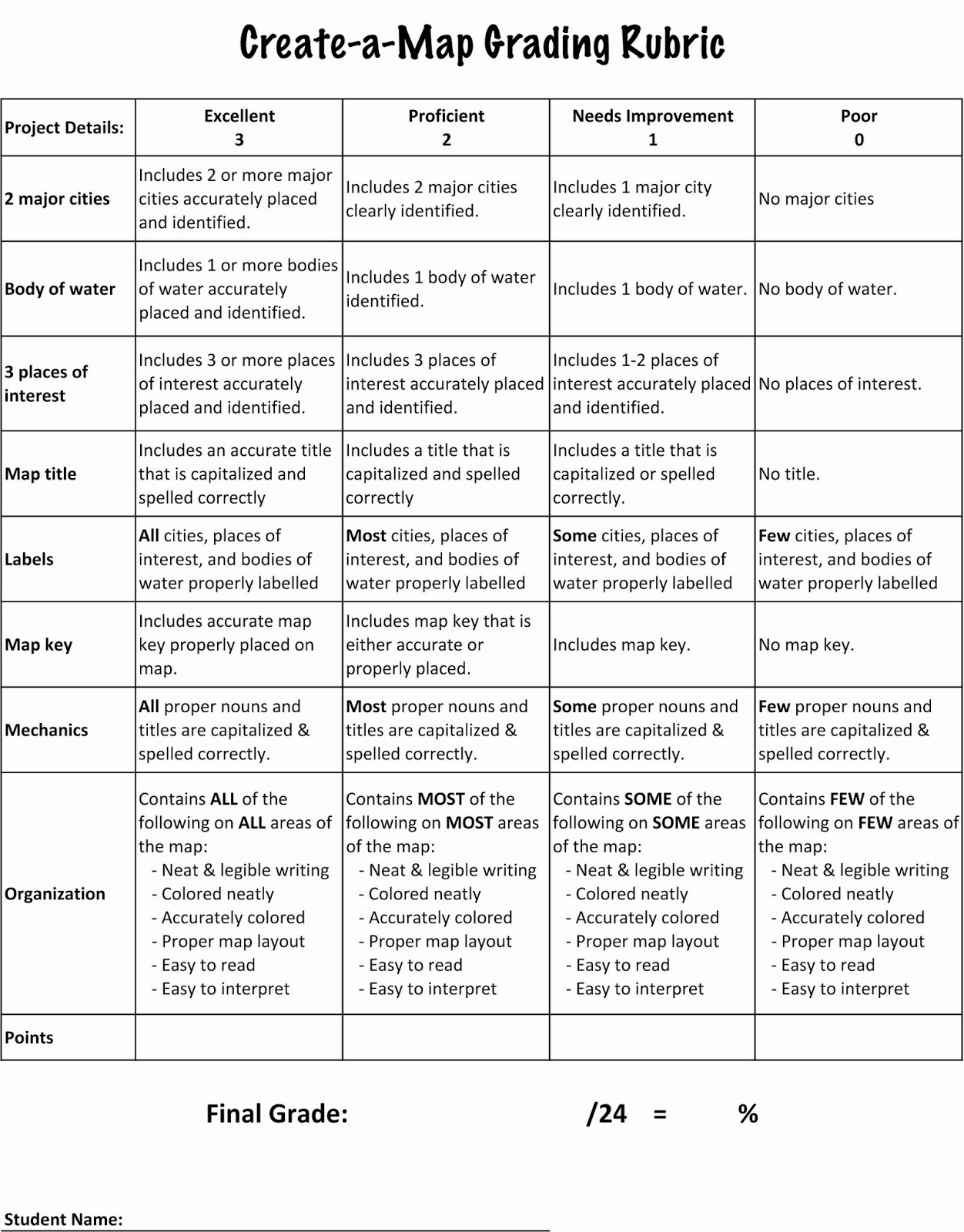
Data, Calculations, and Conclusion - Acid Base Titration Lab.
Sailing on the “C“: Titration with a Twist. Anna-Marie Finger CHEM 1811-Section 12 September 14, 2012 Results and Discussion. In this experiment a solution containing ascorbic acid was titrated with an iodine solution in order to determine the concentration of ascorbic acid in limes, lemons and oranges (eq 1).
Guidance for Writing Lab Reports WRITTEN BY ADAM BEAGLES, STEPHEN BECK, LIZZY CROSS, ANDREW GARRARD AND JEN ROWSON 1 Writing lab reports To write a successful scientific report you need to be clear about what you are trying to. Following the conclusion should be additional, non-essential information.
In this experiment you will use the titration technique to determine the concentration of the provided sodium hydroxide solution. The process of carrying out a titration allows us to accurately.
Writing a powerful conclusion shows your professor that you took your research seriously and immersed yourself in the study. A quality laboratory report ends with an insightful conclusion that convincingly explains why the findings matter. The conclusion is your final opportunity to state your case and back it up with hard evidence.
In conclusion, there was a large discrepancy for the percentage yields, which can be fixed through modifications of the laboratory techniques, but two of the other objectives for this experiment were met.
In this experiment, we shall use acid-base titration to determine the weight-to-volume percentage of acetic acid and ammonia. View Notes - Titration lab from BIO 302 at SUNY Brockport. Uploaded on Oct 16, 2015. by rusha (Teacher). In this experiment titration curves for the following acids and bases will be recorded and analyzed: 1.
Download file to see previous pages In this experiment, the searchers are trying to perform a complex formation reaction for analytical purposes. The main aim of this titration reaction is to determine the presence of calcium ions in a titrant by a method referred to as titrimetric.